
- Apr 8
Innovation and the future of wearable technology
- Rohan S and Ariadne A (edited by Jayden B)
- Biology, Chemistry, Technology
- 0 comments
Wearable technology has changed a lot in the past decade, going from simple fitness trackers to devices that can monitor important health data in real time. What used to require specialised equipment can now be done using small portable devices. This reflects a wide shift in innovative healthcare which focuses more on preventative healthcare and spotting problems early rather than treating them after they happen [1]. It allows for earlier intervention and more personalised care, and a better understanding of how lifestyle and environmental factors influence health. With conditions like cardiovascular disease and diabetes becoming more common, there’s a growing need for tools that people can use easily in their everyday lives [2].
Many wearable devices now include features that move closer to clinical use. Some smartwatches offer electrocardiogram functions that record the electrical activity of the heart. These can help detect irregular rhythms such as atrial fibrillation [3]. Other devices take a different approach. Chest strap monitors such as the Polar H10 Heart Rate Sensor measure electrical signals directly and give more accurate readings during exercise [4]. Smart rings such as the Oura Ring Gen3 focus on long-term trends [5]. They track resting heart rate and heart rate variability to show patterns in recovery and overall cardiovascular health. A major strength of these devices is the amount of data they collect over time. Continuous tracking reveals patterns that single measurements can miss. This includes irregular heart rhythms that occur occasionally or slow changes in resting heart rate [6]. At the same time, there are clear limits to such innovation. Movement and poor sensor contact can reduce accuracy, and external interference can affect readings. The data also often still needs clinical review before it can guide treatment. Even with these limits, wearable cardiovascular monitors continue to improve and are now a common part of both personal health tracking and clinical care [7].
Following on from the theme of continuous monitoring, continuous glucose monitors (CGMs) have now become relatively easy to incorporate into their daily lives, transitioning from the once or twice a day blood pricks – once known as self-monitoring blood glucose (SMBG).[8][9] While these have been the standard for the past decades, SMBG only provide isolated measurements, which means patients critical of their blood glucose levels are unable to capture the glycaemic variability, nocturnal fluctuations and directional trends, limiting the ability to prevent both acute complications e.g. hypo/hyperglycaemia and even long-term sequelae e.g. CVDs, and peripheral neuropathy.[9][10][11][12][13][14] CGMs on the other hands, addresses this directly, producing consistent graphical data, providing a more complete physiological picture, and more thoughtful and proactive interventions, such as understanding which foods raise glucose levels the most, and which the least.[8][9]
CGMs currently utilise enzyme-based amperometric sensors. [15] Amperometry revolves around measuring the electrical current generated as a result of the oxidation or reduction of an electroactive species at a constant potential, wherein the current produced is directly proportional to the concentration of a specific analyte.[16][17] In CGMs, there is a thin filament consisting of 3 layers: biocompatible membrane to ensure no immune rejection, an immobilised enzyme layer of glucose oxidase (GOx), and a platinum electrode. The filament penetrates subcutaneous tissue, usually in the arm, where GOx oxidises glucose in interstitial fluid, forming gluconic acid and hydrogen peroxide.[18][19]
Glucose + Oxygen + Water Gluconic Acid + Hydrogen Peroxide
C6H12O6 + O2 + H2O => C6H12O7 + H2O2
This hydrogen peroxide released now travels across the platinum positive electrode, which then oxidises the hydrogen peroxide to release electrons, thus generating a current.[18][19]
H2O2 => O2 + 2H+ +2e-
Due to the stoichiometric relationship between the release of electrons (current size) and glucose concentration in interstitial fluid, blood glucose concentration can be quantified.[19]
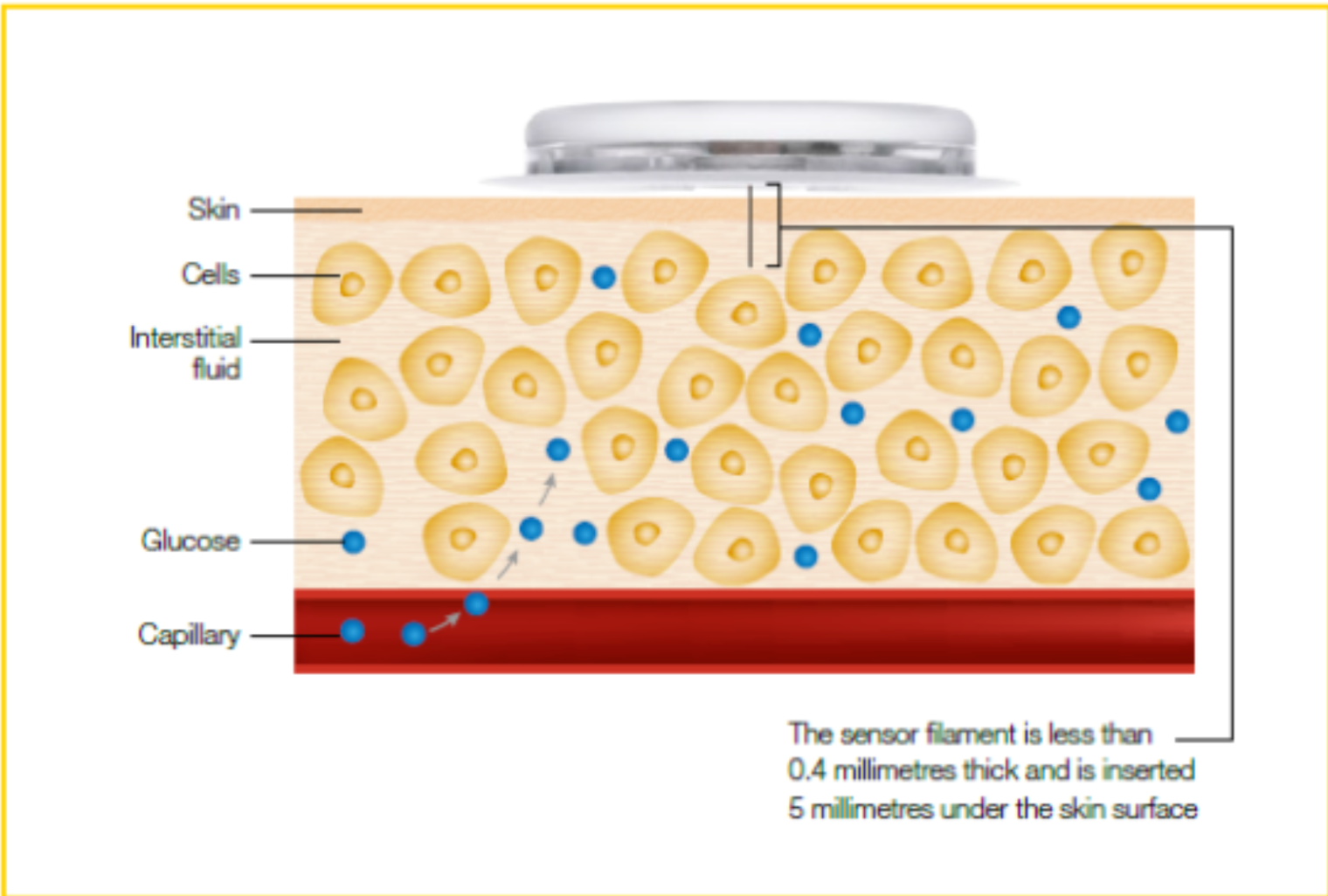
[19]
There have been various studies carried out which have provided significant evidence in the utility of CGMs in clinical environments. For Type I Diabetes, two key studies have been carried out: the DIAMOND study and the GOLD study – do not be alluded by their attractive acronyms, they have more scientific grounding behind them.[20][21][22]
The DIAMOND (Multiple Daily Injections and Continuous Glucose Monitoring in Diabetes) study was an RCT involving 158 Type I Diabetes adults with multiple daily insulin injections, and demonstrated CGM use significantly improved glycaemic control, where over 24 weeks, the group utilising CGM ascertained a 0.6% greater reduction in HbA1c compared to the control group (p<0.001), while also increasing the glucose time-in-range by 9% (this can be approximated to an additional 2 hours per day).[21] HbA1c is glycated haemoglobin, and so due to glucose binding irreversibly to red blood cells, which have a 120-day life span, the rate of HbA1c is directly proportional to average blood glucose concentration, and so a decrease in this is beneficial for patients.[23] Similar results have also been shown in the GOLD (Glucose Optimal Diabetes) with a 0.43% reduction in HbA1c and a 9.5% increase in glucose time-in-range.[22]
For Type II Diabetes, one of the most pivotal studies was the REPLACE (Randomized Evaluation of a Factory-Calibrated Continuous Glucose Monitor in Type 2 Diabetes), which looked at 224 patients who were using intensive insulin therapy, and demonstrated that factory-calibrated CGMs were able to reduce Hb1Ac 0.4% more compared to SMBG, and more notable was patients spent around 40% less time in hypoglycaemia with CGMs, highlighting thus their efficacy for both types of Diabetes.[24]
While CGMs are evidently beneficial to many people trying to make the conscious effort to monitor their blood glucose levels, there are a few limitations, most importantly, the lag phenomenon.[25][26][27] As explained and visually represented above, substances on CGM filaments are only reacting to glucose in the interstitial fluid, not directly in the blood. This means during rapid glucose changes e.g. eating a meal, or an insulin dose, the CGM recording will have an 8–12-minute lag often.[25][26][27] Apart from this, other limitations include calibration drift and sensor degradation after 10-14 days (at which point replacement is necessary), as well as endogenous and exogenous substances interfering with the electrochemical balance, such as salicylic acid, or ascorbic acid (Vitamin C).[27]
So far, the wearable innovations we have covered share a common functionality – both collected metabolic data to inform a user or clinician, acting as merely a diagnostic aid for further decision making. However, there are other means of modern medical innovations, focusing beyond continuous monitoring to direct intervention. Most notably, transcutaneous electrical nerve stimulation (TENS) devices e.g. Livia, represent this alternative paradigm, shifting from biomarker tracking to pain modulation through electrical stimulation.[28]
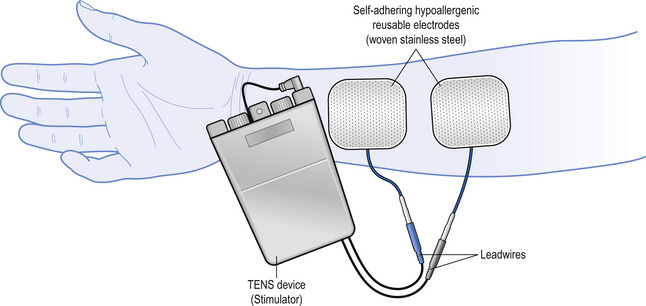
Pain – one of the most common reasons for medical consultations – affects millions of people worldwide.[29] To focus on a specific use case of TENS, we will look at primary dysmenorrhea, which is menstrual pain, without any underlying pelvic pathology as such, and affects over half of menstruating women.[30] This hence represents a clear burden on quality of life of many women, so conventional management has often been centred around hormonal contraceptives and non-steroidal anti-inflammatory drugs (NSAIDs) e.g. Ibuprofen.[30][31] However, both have inherent flaws: hormonal contraceptives may go against personal views or creating a hormonal imbalance with unprecedented side effects e.g. prolonged nausea, fatigue, muscle pain; NSAIDs have been linked with gastrointestinal (GI) irritation, renal toxification and a CVD risk factor.[31] Hence, for primary dysmenorrhea, TENS e.g. brand Livia is a growing alternative, which incorporates a non-invasive, drug-free, wearable device which relieves menstrual pain through the electrostimulation of peripheral nerves.[28][32][33]
To understand TENS, first we must look into physiological basis for it: the Gate Control Theory (GCT), proposed by Melzack and Walls in 1965, which broke previous conceptions around pain being a linear transmission from peripheral nociceptors to the brain.[34][35] Pain signals originate from nociceptors, which are specialised sensory neurons that detect any (potentially) damaging stimuli, and are free nerve endings along 2 types of axons: small, lightly myelinated, Aδ fibres for transmitting sharp, acute, well-localised pain, and smaller, unmyelinated, C fibres for transmitting dull, chronic, aching pains. In contrast, it is important to note large, heavily myelinated, Aβ fibres transmit non-painful sensory information.[34][35]
The former 2 axon types transmit pain signals to the spinal cord, where within the dorsal horn, in a specific region known as the substantia gelatinosa, where they undergo modulation by inhibitory interneurons, which act as gates, dictating whether the electrical signal goes to the brain.[34][35] If there is predominantly Aδ or C fibre activity, the gate remains open to transmit pain signals to the brain, but if it is predominantly Aβ fibre activity, the gate is closed, and pain transmission to the brain is inhibited, preventing pain perception even if there is nociceptive stimulation.[34][35]
Now that the theory is covered, how does TENS capitalise on this neurological mechanism? TENS delivers low-voltage electrical pulses through electrodes to the skin, which recruits Aβ fibres, overwhelming the substantia gelatinosa with non-painful sensory input.[33][35] The net effect: reduction in perceived pain, without necessarily reducing nociceptive stimulation.[33] For menstrual cramp relief specifically, the electrodes are placed over the suprapubic region, targeting T10–L1 spinal segments – dermatomes corresponding to uterine innervation.[33][36] Furthermore, the electrical signals generated are biphasic pulses, that is, electrical signals alternating between positive and negative phases, to ensure there is a minimised net charge accumulation at the electrode-skin interface, reducing risk of skin irritation and electrochemical burns.[33][37]
Though this is a relatively new intervention, there have been high-quality studies into this use case of TENS. A highly regarded example would be a Cochrane Systematic Review, where over 20 RCTs were carried out on 585 women, and provided significant evidence that for both high and low frequency TENS, there was significant reductions in menstrual pain compared to placebo (mean differences of 1.39 and 2.04 on a 0–10 scale, respectively).[38][39] Livia, a branded FDA-approved TENS device, also carried out a study where a randomised, double-blind trial of 65 women with primary dysmenorrhea demonstrated significant reduction in pain scores of active TENS compared to a placebo. This, alongside other research, led to Livia’s 510(k) clearance from the FDA.[40]
While TENS are clearly beneficial for women dealing with primary dysmenorrhea, and being NHS-recommended and FDA approved, there are still a few problems and questionability around the true efficacy of TENS. Pivotal limitations include the heterogeneity of the studies carried out, with very varied stimulation patterns and placebo controls. Furthermore, effective blinding was rarely carried out on all of these studies, such as in the Cochrane review, 13 out of the 20 studies has inadequate blinding or were simply unblinded, perhaps skewing the significance of the results.[39]
On the physiological side, while it is true that the Gate Theory Model holds reasonably well to evidence these results, there are added nuances which are not yet fully understood, one of which suggests that high-frequency and low frequency TENS operate through different mechanisms, the former through gate control, whereas the latter with opioid-mediated descending inhibition, and even this distinction is not absolute.[34] Clinically, there are varying degrees of responsiveness to the treatment, and the placebo effect even accounting for a large proportion of responses, so the physiological effects can often be difficult to determine.[43] Practically, there is an obvious learning curve when it comes to frequency setting and placement of the device itself, as mentioned above if not placed in the suprapubic region properly, will not work effectively as such. Moreover, there are a few contraindications in several populations, such as pregnant women, women with implanted electronic devices e.g. pacemakers, and women prone to epilepsy, limiting its usage.[34][43][44]
Wearable medical technology is shifting healthcare towards prevention and continuous monitoring. Devices like cardiovascular monitors and CGMs provide valuable real-time data, while innovations such as TENS show that wearables can also play a role in treatment. Despite clear benefits, limits such as accuracy issues and physiological delays mean these devices should support and not replace the important clinical judgement. As technology improves, wearable healthcare is very likely to become an increasingly important part of modern healthcare.
Bibliography
Kang HS, Exworthy M. Wearing the Future—Wearables to Empower Users to Take Greater Responsibility for Their Health and Care: Scoping Review. JMIR mHealth and uHealth [Internet]. 2022 Jul 13;10(7). Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC9330198/
Edelmann S. Wearable technologies in healthcare [Internet]. Diagnostics. Roche Diagnostics International; 2025. Available from: https://diagnostics.roche.com/global/en/healthcare-transformers/article/wearable-technology-in-healthcare.html
Lally C, Narain V. Consumer wearable devices and disease prevention [Internet]. POST. 2025. Available from: https://post.parliament.uk/research-briefings/post-pn-0741/
Roy M. The Best Heart Rate Monitors for Every Athlete (2026 Guide) [Internet]. North Sports. 2025 [cited 2026 Mar 24]. Available from: https://northsports.co.uk/blogs/performance-tech/the-best-heart-rate-monitors-for-every-athlete-2026-guide
What’s the Point of Smart Rings? The Real Value Beyond the Hype [Internet]. RingConn. 2025. Available from: https://ringconn.com/blogs/news/smart-ring-real-value-and-purpose
Vijay Kumar Malesu. News-Medical [Internet]. News-Medical. 2025. Available from: https://www.news-medical.net/health/How-Wearable-Tech-Is-Reshaping-Preventative-Health.aspx
Kleinman Z. Wearable tech can monitor our health but why are doctors so sceptical? BBC News [Internet]. 2024 Dec 2; Available from: https://www.bbc.co.uk/news/articles/c79zpzdv4vn
Continuous glucose monitors [Internet]. Diabetes UK. 2025. Available from: https://www.diabetes.org.uk/about-diabetes/looking-after-diabetes/technology/continuous-glucose-monitors
Basics of Self Monitoring of Blood Glucose in Multiple Daily Injections (SMBG) | The Association of British Clinical Diabetologists [Internet]. Abcd.care. 2026 [cited 2026 Mar 24]. Available from: https://abcd.care/resource/current/basics-self-monitoring-blood-glucose-multiple-daily-injections-smbg
NHS . Continuous glucose monitoring and hybrid closed loop for diabetes [Internet]. nhs.uk. 2024. Available from: https://www.nhs.uk/tests-and-treatments/cgm-and-hcl-for-diabetes/
Suh S, Kim JH. Glycemic Variability: How Do We Measure It and Why Is It Important? Diabetes & Metabolism Journal [Internet]. 2015;39(4):273. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4543190/
Institute for Quality and Efficiency in Health Care. Hyperglycemia and Hypoglycemia in Type 2 Diabetes [Internet]. National Library of Medicine. Institute for Quality and Efficiency in Health Care (IQWiG); 2023. Available from: https://www.ncbi.nlm.nih.gov/books/NBK279510/
Sequelae: Definition, Examples, COVID-19, and More | Osmosis [Internet]. www.osmosis.org. Available from: https://www.osmosis.org/answers/sequelae
National Institute of Neurological Disorders and Stroke. Peripheral Neuropathy | National Institute of Neurological Disorders and Stroke [Internet]. www.ninds.nih.gov. 2024. Available from: https://www.ninds.nih.gov/health-information/disorders/peripheral-neuropathy
Miller EM. Using Continuous Glucose Monitoring in Clinical Practice. Clinical Diabetes [Internet]. 2020 Dec;38(5):429–38. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7755046/
Zadel AH, Chiampas K, Maktaz K, Keller JG, O’Gara KW, Vargas L, et al. Continuous Glucose Monitoring in Primary Care: Multidisciplinary Pilot Implementation Study. JMIR Diabetes [Internet]. 2025 Jun 18;10:e69061–1. Available from: https://diabetes.jmir.org/2025/1/e69061
Harvey D. 11.4: Voltammetric and Amperometric Methods [Internet]. Chemistry LibreTexts. 2019. Available from: https://chem.libretexts.org/Bookshelves/Analytical_Chemistry/Analytical_Chemistry_2.1_(Harvey)/11%3A_Electrochemical_Methods/11.04%3A_Voltammetric_and_Amperometric_Methods
Dungan K, Verma N. Monitoring Technologies – Continuous Glucose Monitoring, Mobile Technology, Biomarkers of Glycemic Control [Internet]. Nih.gov. MDText.com, Inc.; 2023. Available from: https://www.ncbi.nlm.nih.gov/books/NBK279046/
Lee I, Probst D, Klonoff D, Sode K. Continuous glucose monitoring systems - Current status and future perspectives of the flagship technologies in biosensor research -. Biosensors and Bioelectronics. 2021 Jun;181:113054.
Manov AE, Chauhan S, Dhillon G, Dhaliwal A, Antonio S, Donepudi A, et al. The Effectiveness of Continuous Glucose Monitoring Devices in Managing Uncontrolled Diabetes Mellitus: A Retrospective Study. Cureus [Internet]. 2023 Jul 27;15(7):e42545. Available from: https://www.cureus.com/articles/160290-the-effectiveness-of-continuous-glucose-monitoring-devices-in-managing-uncontrolled-diabetes-mellitus-a-retrospective-study#
Beck RW, Riddlesworth T, Ruedy K, Ahmann A, Bergenstal R, Haller S, et al. Effect of continuous glucose monitoring on glycemic control in adults with type 1 diabetes using insulin injections: The DIAMOND randomized clinical trial. JAMA. 2017;317(4):371–8.
Lind M, Polonsky W, Hirsch IB, Heise T, Bolinder J, Dahlqvist S, et al. Continuous Glucose Monitoring vs Conventional Therapy for Glycemic Control in Adults With Type 1 Diabetes Treated With Multiple Daily Insulin Injections. JAMA [Internet]. 2017 Jan 24;317(4):379. Available from: https://jamanetwork.com/journals/jama/fullarticle/2598771
Eyth E, Zubair M, Naik R. Hemoglobin A1C [Internet]. PubMed. Treasure Island (FL): StatPearls Publishing; 2023. Available from: https://www.ncbi.nlm.nih.gov/books/NBK549816/
Aleppo G, Ruedy KJ, Riddlesworth TD, Kruger DF, Peters AL, Hirsch I, et al. REPLACE-BG: A Randomized Trial Comparing Continuous Glucose Monitoring With and Without Routine Blood Glucose Monitoring in Adults With Well-Controlled Type 1 Diabetes. Diabetes Care. 2017 Feb 16;40(4):538–45.
Karakuş KE, Sakarya S, Yeşiltepe Mutlu G, Berkkan M, Muradoğlu S, Can E, et al. Benefits and Drawbacks of Continuous Glucose Monitoring (CGM) Use in Young Children With Type 1 Diabetes: A Qualitative Study From a Country Where the CGM Is Not Reimbursed. Journal of Patient Experience [Internet]. 2021 Jan;8(1):237437352110565. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC8646182/
Researchers warn continuous glucose monitors can overestimate blood sugar levels [Internet]. Bath.ac.uk. 2025. Available from: https://www.bath.ac.uk/announcements/researchers-warn-continuous-glucose-monitors-can-overestimate-blood-sugar-levels/
Oriot P, Klipper dit kurz N, Ponchon M, Weber E, Colin IdesM, Philips JC. Benefits and limitations of hypo/hyperglycemic alarms associated with continuous glucose monitoring in individuals with diabetes. Diabetes Epidemiology and Management. 2023 Jan;9(9):100125.
NHS website. TENS (transcutaneous electrical nerve stimulation) [Internet]. nhs.uk. 2017. Available from: https://www.nhs.uk/tests-and-treatments/transcutaneous-electrical-nerve-stimulation-tens/
Wager TD. Managing Pain. Cerebrum: the Dana Forum on Brain Science [Internet]. 2022 Mar;2022:cer-03-22. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC9224345/
Itani R, Soubra L, Karout S, Rahme D, Karout L, Khojah HMJ. Primary Dysmenorrhea: Pathophysiology, Diagnosis, and Treatment Updates. Korean Journal of Family Medicine [Internet]. 2022 Mar 17;43(2):101–8. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8943241/
Period pain (dysmenorrhoea) [Internet]. NHS inform. Available from: https://www.nhsinform.scot/healthy-living/womens-health/girls-and-young-women-puberty-to-around-25/periods-and-menstrual-health/period-pain-dysmenorrhoea/
Teoli D, An J. Transcutaneous Electrical Nerve Stimulation (TENS) [Internet]. Nih.gov. StatPearls Publishing; 2019. Available from: https://www.ncbi.nlm.nih.gov/books/NBK537188/
Elboim-Gabyzon M, Kalichman L. Transcutaneous Electrical Nerve Stimulation (TENS) for Primary Dysmenorrhea: An Overview. International Journal of Women’s Health. 2020 Jan;Volume 12:1–10.
Physiopedia. Gate Control Theory of Pain [Internet]. Physiopedia. 2024. Available from: https://www.physio-pedia.com/Gate_Control_Theory_of_Pain
Braz J, Solorzano C, Wang X, Basbaum Allan I. Transmitting Pain and Itch Messages: A Contemporary View of the Spinal Cord Circuits that Generate Gate Control. Neuron. 2014 May;82(3):522–36.
Dermatomes: What They Are & Locations [Internet]. Cleveland Clinic. 2022. Available from: https://my.clevelandclinic.org/health/body/24379-dermatomes
Hingne PM, Sluka KA. Differences in Waveform Characteristics Have No Effect on the Anti-Hyperalgesia Produced by Transcutaneous Electrical Nerve Stimulation (TENS) in Rats With Joint Inflammation. The Journal of Pain. 2007 Mar;8(3):251–5.
Han S, Kyoung Sun Park, Lee H, Kim E, Zhu X, Jin Moo Lee, et al. Transcutaneous electrical nerve stimulation (TENS) for pain control in women with primary dysmenorrhoea Overview. Cochrane library. 2024 Jul 22;2024(7).
Han S, Kyoung Sun Park, Lee H, Kim E, Zhu X, Jin Moo Lee, et al. Transcutaneous electrical nerve stimulation (TENS) for pain control in women with primary dysmenorrhoea. Cochrane library. 2024 Jul 22;2024(7).
mylivia.com [Internet]. mylivia.com. 2026 [cited 2026 Mar 24]. Available from: https://mylivia.com/?srsltid=AfmBOoqQqujuUFFzeEYFWvYjsXKNokt4Hnc0VxIakshons1UsSr_keFg
Ltd L, Wiesen I. 2020 [cited 2026 Mar 24]. Available from: https://www.accessdata.fda.gov/cdrh_docs/pdf18/K183110.pdf
Clinicaltrials.gov. 2026 [cited 2026 Mar 24]. Available from: https://clinicaltrials.gov/study/NCT03064945?tab=results
Rodrigues JC, Avila MA, Driusso P. Transcutaneous electrical nerve stimulation for women with primary dysmenorrhea: Study protocol for a randomized controlled clinical trial with economic evaluation. Rosier PFWM, editor. PLOS ONE. 2021 May 20;16(5):e0250111.
NHS website. TENS (transcutaneous electrical nerve stimulation) [Internet]. nhs.uk. 2017. Available from: https://www.nhs.uk/tests-and-treatments/transcutaneous-electrical-nerve-stimulation-tens/
Image links
https://th.bing.com/th/id/OIP.YgPybddfkhk8w_ljs3VKrwHaEK?w=324&h=182&c=7&r=0&o=7&dpr=2&pid=1.7&rm=3
https://th.bing.com/th/id/R.e664e105cb2b5dfbe9dcac10bc872dd0?rik=RRpnkFvubHtLzw&pid=ImgRaw&r=0