
- Sep 6, 2024
The Future of Fertility: Can We Extend Women's Biological Clocks?
- Giselle R and Jenna M
- Biology
- 0 comments
As we navigate a modern world, the importance of career advancement overpowers the development of family life. This has resulted in the total fertility rate (TRF) in England and Wales to decrease from 1.55 children per woman in 2021 to 1.49 in 2022. Since 2010 the UK has seen a decline in TFR across all age groups except for the twenties (Munro & Lyons, 2024). Traditionally, fertility peaks during a woman’s early twenties and gradually declines until reaching menopause at around 50, where it is impossible to naturally reproduce. The difficulty to conceive as a woman ages, otherwise known as the “biological clock” was coined by Richard Cohen in 1978 (Healey, 2014) to describe the dilemma facing the population. However, genome-editing technology such as CRISPR may be the catalyst to extending a woman’s fertility window, offering opportunities to focus on life aspirations without compromising motherhood.
What is CRISPR and How Does it Work?
CRISPR, stands for Clustered Regularly Interspaced Short Palindromic Repeats, is a revolutionary bacterial system that can be programmed to edit DNA in particular locations. Essentially, it is a way to modify genes in the living cells of virtually all species. Originally discovered by Francisco Mojica, it was later developed by Feng Zang, Jennifer Doudna, and Emmanuelle Charpentier to edit mice cells and human cells (Rodríguez Fernández, 2022). CRISPR works by targeting a specific chain of genetic code and producing an enzyme which will bind to the DNA sequence and produce a double strand break; this shuts the gene off with unprecedented accuracy. However, by modifying Cas9–one of the enzymes released—the gene expression won’t be cut and will instead insert a new sequence during the cell’s natural repair process, this includes: protein tags, mutations, and loxP sites (Cho, et al., 2018). A loxP site is a sequence that occurs naturally in the genome of certain bacteriophages which is necessary for development and replication. By inserting this, it allows for the recombination of the gene, enabling it to be ‘knocked out’. Within mice, this is the workflow of CRISPR-Pro knockout:
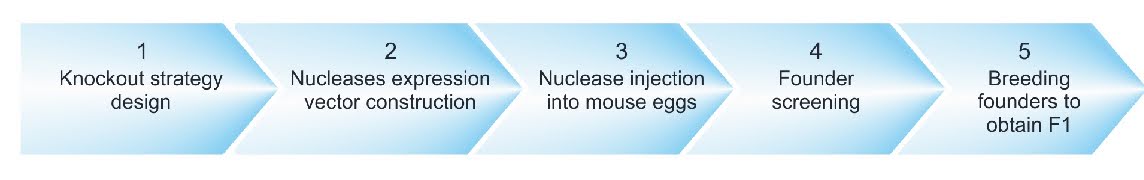
Fig 1: (Cyagen, n.d)
The final product is a genetically modified litter which bears the knockout allele. Since mice share approximately 95% of their DNA with humans, CRISPR is capable of modifying “typos” in the three-billion-letter sequence of the human genome to correct debilitating diseases. (Verma, 2020).
A Success Story: How Victoria Gray Overcame Sickle Cell with CRISPR
Sickle cell disease is an inherited disorder which inhibits the ability of hemoglobin to carry oxygen; since they are malformed it causes blockages in the blood vessels. For Victoria Gray this meant that she experienced “pain that felt like lightning strikes and sometimes being hit by a truck”, as she said during her interview with the CF Foundation, (Cystic Fibrosis Foundation, 2023) that it also greatly altered her family life with her children fearing that everyday could be her last. After relentless treatment in hospital with blood transfusions and IV’s, she received an experimental CRISPR-Cas9 therapy. Cells were removed from her bone marrow which were then edited by CRISPR and infused into her body. This grueling procedure took months, but subsequently, she was able to take an active role in her children’s lives, travel across the world with her husband, work, and no longer worry about sudden attacks which could lead to an early grave. Despite all of this, there’s a divide between experts: some praise the breakthrough since it allows for embryonic screening to avoid lethal diseases and currently cures anemia, sickle cell disease, and blindness; contrastingly, others believe that it may create ‘designer babies’ in the near future and ethical concerns of bias against inherited disorders. However, Victoria Gray told CNN (Hunt, 2023), “I’m here really to be a light because there’s mixed feelings about gene editing. And I think people can see the positive result of it. You know that a person who was once suffering in life, was miserable, now is able to be part of life and enjoy it.” With a 77% success rate of disease control (Henderson, 2023), it’s an undeniable fact that CRISPR technology has the potential to improve countless lives and revolutionize the medical industry.
How Might CRISPR Be Used to Extend Fertility and Improve Egg Health?
As women age, egg quality deteriorates, resulting in a higher incidence of chromosomal abnormalities such as genetic defects or miscarriages. Several factors contribute to this: decreased egg quantity, hormonal change due to irregularity, change in uterine environment, accumulated DNA damage, and mitochondrial DNA (mtDNA) abnormalities. DNA damage can result in apoptosis, leading to cell inflammation and then loss of homeostasis within the gene, thus causing aging in a cell. In addition to this, aging can occur with the accumulation of mutations which also affect the regulation of optimum levels (Vijg, 2021). So, by designing an RNA molecule that aligns with the mutated DNA sequence, and then combining it with Cas9 –the 160 kilodalton protein- it can “cut through the DNA like sharp scissors” according to Qi, a former student at Doudna’s lab (Shwartz, 2018).
Another factor often ignored in the decline of oocyte quality is mtDNA damage as the mitochondria is crucial for maintaining the health of cytoplasm; which supplies the majority of nutrients for the zygote (Wu & Zhang, 2023). Aging in the uterine environment is characterized by diminished ovarian reserve, which can also be referred to as primary ovarian insufficiency (POI). It is suggested that low mtDNA content is associated with the impaired oocyte quality observed in ovarian insufficiency (Chrétien, et al., 2005). When it is deficient, the cells cannot produce the necessary proteins for effective oxidative phosphorylation (OXPHOS), resulting in the cell’s inability to produce the ‘energy currency’. However, this can be rectified by repopulating mitochondria with wt-mtDNA (wild type mtDNA). Once accomplished the mitochondria can perform OXPHOS effectively (Hjort, et al., 2022). Since mitochondria is a crucial organelle for any cell, once the damage is altered a leading cause for diminished fertility is gone. Therefore, there is a potential to extend the viability of egg cells and improve entire populations.
The Socioeconomic Consequences of an Aging Population
Countries in Stage 5 of the Demographic Transition Model (DTM) are classified by an aging population that has an exceeding death rate in comparison to the birth rate. Some examples of this are: Germany, Greece, Estonia, Croatia, Ukraine, and Portugal (World Population Review, 2024). If this trend across Europe continues, England will encounter the same complications.
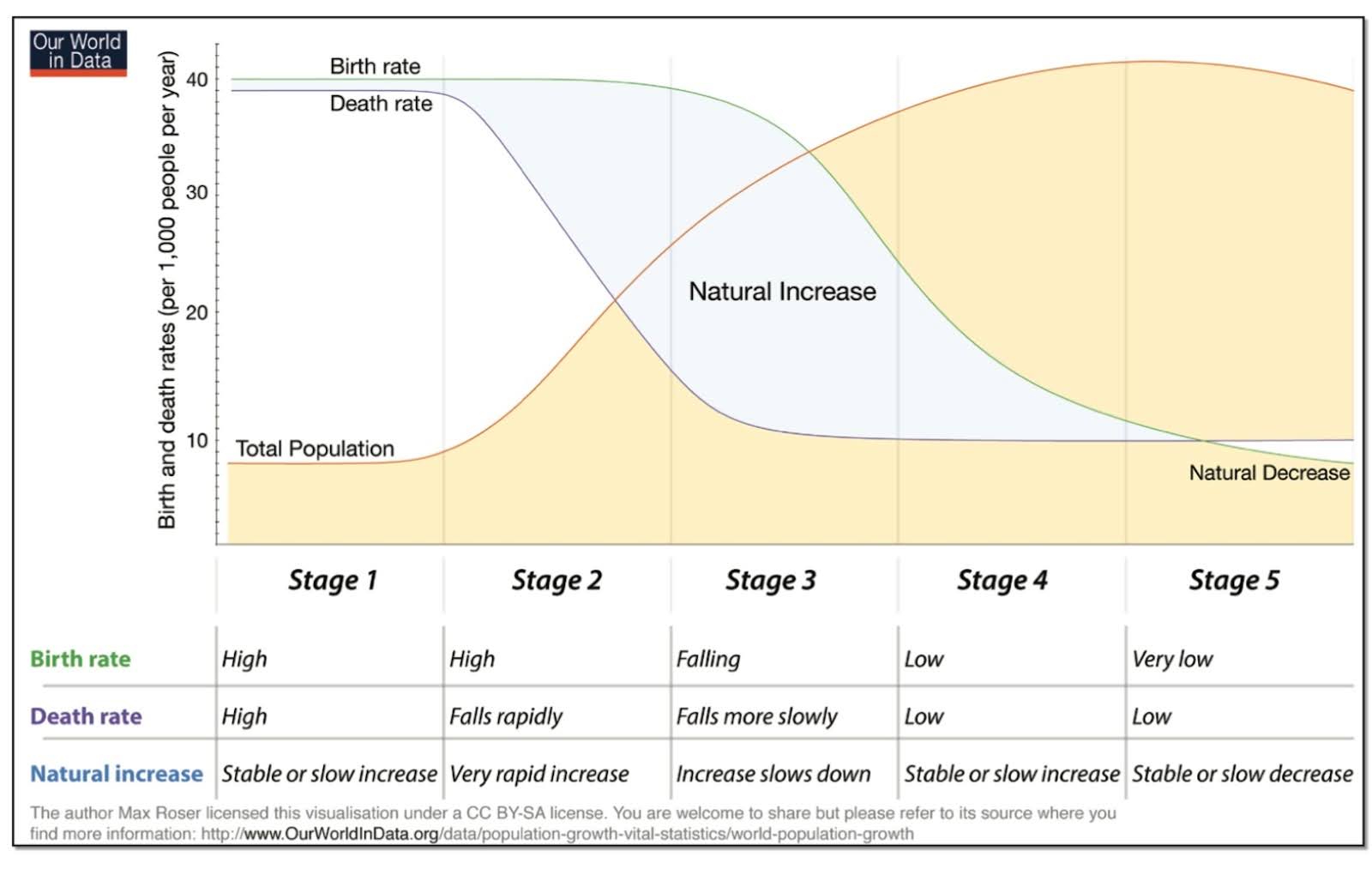
Fig 2: (Roser, n.d)
Graying populations are indicative of a reduced number of people in their childbearing years, causing a strain on health services as the elderly often suffer from comorbidity, a burden on caregivers affecting intergenerational relationships, higher government spending on retirement funds, and a decline in the working-age population. In some instances, this results in wage inflation and a vicious cycle of price spiral. This is due to the increased dependency placed onto the smaller pool of workers in order to collect tax for health costs, pension benefits, and other publicly funded programs (Borji, 2023). This could devastate England’s already damaged economy after its investment in the Brexit Referendum, exacerbated by the lack of migration and COVID-19’s effects, undoubtedly deteriorating the economy. (Goss, 2024) Potentially, this could also contribute to the major isolation crisis that the elderly face, with 1.4 million older people in the UK feeling lonely (AgeUK, 2021).
A larger workforce would not only support the economy but also allow for a more equitable distribution of aid for the older generation. If women from 50-60 had the ability to produce healthy offspring, there would be a steadier replenishment of the workforce; ensuring a constant influx of young workers. This would lead to higher productivity and long-term economic growth since industries would benefit from new talent and consumers. What’s more, the government would face less pressure when putting tax payers’ money into pensions, allowing funding to instead go to first responders, education, or important infrastructure.
The New ‘Modern’ Woman
By increasing the window of time women can conceive, there will still be opportunities for career advancement or education whilst reducing the pressure to foster a stable family. Some millennial women believe that “if and when they have kids, it will be harder for them to advance in their careers”, a sentiment supported by the 59% of women surveyed who believe that being a working parent is a significant barrier (Pew Research Centre, 2013). Thus, the ability to balance personal and professional attainment becomes achievable. By addressing these challenges, if further explored gene-editing technology, despite currently showing normal fertility levels in mice (Abbasi, 2022), presents promising solutions to stabilize demographic trends and support social well-being.
Ultimately, extending the ‘biological clock’ through CRISPR is not about devaluing early motherhood but instead expanding choice without forcing difficult trade-offs. Choosing a career over a young maternal path should not be considered a derision. Motherhood should not be a burden.
References:
Abbasi, F., Emori, C., Fujihara, Y., Ikawa, M., Kiyozumi, D., Kodani, M., & Mori, M. (2020, March 28). CRISPR/Cas9-mediated genome editing reveals Oosp family genes are dispensable for female fertility in mice. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7226750/
Borji, H. S. (2023, December 11). 4 global economic issues of an aging population. https://www.investopedia.com/articles/investing/011216/4-global-economic-issues-aging-population.asp
Cho, A., Cho, K., Hall, B., Khillan, J., Kulkarni, A. B., & Limaye, A. (2018, September 4). Genome editing in mice using CRISPR/Cas9 technology. https://currentprotocols.onlinelibrary.wiley.com/doi/10.1002/cpcb.57
Chrétien, M. F., Jacques, C., Malthièry, Y., May-Panloup, P., Reynier, P., & Vasseur, C. (2005, March 1). Low oocyte mitochondrial DNA content in ovarian insufficiency. https://academic.oup.com/humrep/article/20/3/593/
Cystic Fibrosis Foundation. (2023, November 4). CF Foundation | Victoria Gray’s sickle cell gene editing story [Video]. YouTube. https://youtu.be/Qfk51dt0iVk?si=9etHuvEuMX_xk8Gf
DTM Stage 5 Countries 2024. (2024). World Population Review. https://worldpopulationreview.com/country-rankings/dtm-stage-5-countries
Goss, L. (2024, February 12). Brexit has damaged U.K. economy with higher inflation and much weaker growth, Goldman Sachs says in new study. https://www.marketwatch.com/story/higher-inflation-much-weaker-growth-brexit-has-hit-the-u-k-economy-hard-says-goldman-sachs-e6d1211e
Healey, J. (2014, October 10). Rejecting the biological clock.
https://isps.yale.edu/news/blog/2014/10/rejecting-the-biological-clock
Henderson, H. (2023, March 17). CRISPR clinical trials: A 2023 update. https://innovativegenomics.org/news/crispr-clinical-trials-2023/
Hjort, M., Larsson, J., Oburoglu, L., Schmiderer, L., & Yudovich, D. (2022, November 4). Site-specific CRISPR-based mitochondrial DNA manipulation is limited by gRNA import. https://www.nature.com/articles/s41598-022-21794-0
Hunt, K. (2023, March 9). How human gene editing is moving on after the CRISPR baby scandal. https://amp.cnn.com/cnn/2023/03/09/health/genome-editing-crispr-whats-next-scn
Loneliness Research and Resources. (2021, December). AgeUK. https://www.ageuk.org.uk/our-impact/policy-research/loneliness-research-and-resources/
Munro, M., & Lyons, M. (2024, February 23). Births in England and Wales: 2022 (refreshed population). https://www.ons.gov.uk/peoplepopulationandcommunity/birthsdeathsandmarriages/livebirths/bulletins/birthsummarytablesenglandandwales/2022refreshedpopulations
On Pay Gap, Millennial Women Near Parity – For Now. (2013, December 11). Pew Research Centre. https://www.pewresearch.org/social-trends/2013/12/11/on-pay-gap-millennial-women-near-parity-for-now/
Figure 1: Point Mutation Mice. (n.d.). Cyagen. https://www.cyagen.com/us/en/service/crispr-based-genome-editing-point-mutation-mice.html
Rodríguez Fernández, C. (2022, June 23). Francis Mojica, the Spanish scientist who discovered CRISPR. https://www.labiotech.eu/interview/francis-mojica-crispr-interview/
Figure 2: Roser, M. (n.d.). Budding geographers. https://www.buddinggeographers.com/demographic-transition-model-dtm/
Shwartz, M. (2018, February 26). Target, delete, repair. https://stanmed.stanford.edu/crispr-for-gene-editing-is-revolutionary-but-it-comes-with-risks/
Verma, A. (2020, December 14). Crispy tech: CRISPR-Cas9. https://medium.com/rakt-community/crispy-tech-crispr-cas-9-51275985d0cc
Vijg, J. (2021, July). From DNA damage to mutations: All roads lead to aging. https://www.sciencedirect.com/science/article/abs/pii/S1568163721000635
Wu, F., & Zhang, W. (2023, March 17). Effects of adverse fertility-related factors on mitochondrial DNA in the oocyte: A comprehensive review. https://rbej.biomedcentral.com/articles/10.1186/s12958-023-01078-6